MYATT ATT: Unlocking the Power of Inclusive Digital Therapies for Neurofibromatosis Type 1
MYATT ATT: Unlocking the Power of Inclusive Digital Therapies for Neurofibromatosis Type 1
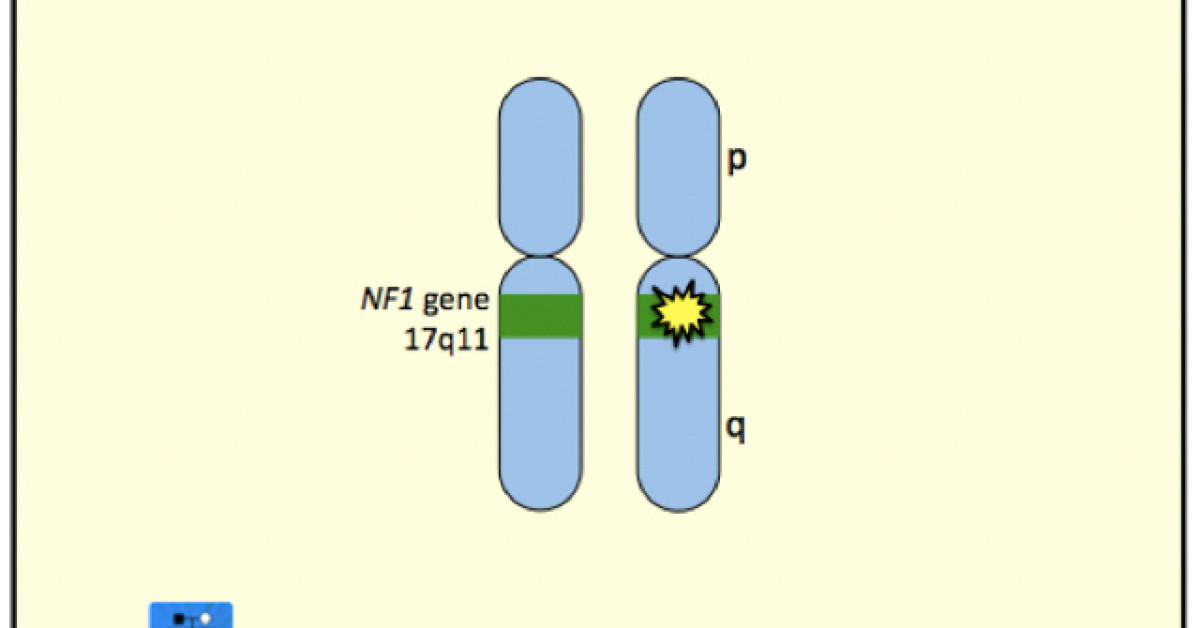
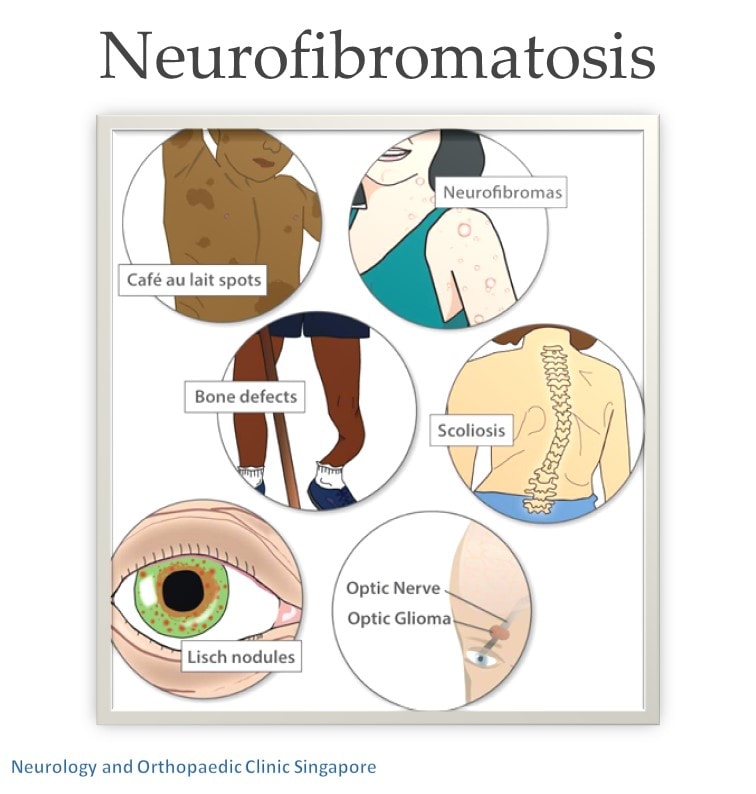
The Myatt Att technology, a wearable brain stimulation device, is revolutionizing the treatment approach for Neurofibromatosis Type 1 (NF1), a genetic disorder affecting nerve tissue development. NF1 patients face a range of debilitating symptoms, including learning disabilities, attention deficits, and emotional struggles, due to the disease's impact on the nervous system. Conventional treatment options have proven to be limited in addressing the complex nature of NF1's symptoms. However, the Myatt Att device has shown promising results in providing a safe, personalized, and effective therapy for patients with NF1, addressing both the cognitive and emotional challenges associated with the disease.
Developed by Silo Neuro Dynamic, Myatt Att uses transcranial alternating current stimulation (tACS) to target and regulate abnormal brain activity patterns in NF1 patients, which contribute to various cognitive, emotional, and behavioral issues. According to Dr. Maria Rodriguez, lead researcher on the project, "NF1 patients often suffer from diffuse and multifaceted symptoms. The Myatt Att technology provides a unique opportunity to tailor the treatment plan to each patient's specific needs, thereby offering a more targeted and efficient approach to alleviating their symptoms." By addressing the underlying brain activity associated with NF1, Myatt Att has the potential to improve the lives of thousands of patients worldwide.
The Science Behind Myatt Att
Myatt Att is a small, non-invasive, and comfortable-to-wear device that is designed to provide transcranial alternating current stimulation. The device is worn on the forehead and uses electroencephalography (EEG) to detect abnormal brain activity in NF1 patients. This information is then used by the Myatt Att algorithm to create a customized treatment plan for each user, aimed at restoring normal brain function and alleviating symptoms.
The Personalized Approach to NF1 Treatment
Unlike traditional treatments that follow a one-size-fits-all approach, Myatt Att takes a highly personalized approach to NF1 treatment, focusing on the unique needs and characteristics of each patient. Prior to using the device, patients undergo a thorough assessment to identify which areas of the brain are vulnerable to abnormal activity. The Myatt Att device then directly targets these areas, providing a tailored electrical stimulation to promote correct brain function.
In a study involving patients with NF1, Dr. Rodriguez saw significant reductions in symptoms of attention and executive functioning, as well as significant improvements in emotional regulation. Another aspect crucial to the Myatt Att device is its adaptability. "The device allows us to easily adjust treatment parameters based on the patient's response," Dr. Rodriguez states. Using this method, clinicians can ensure that patients continue to benefit maximally from treatment.
The Impact of Myatt Att on NF1 Symptoms
Research has demonstrated the efficacy of Myatt Att in treating a range of symptoms associated with NF1, from attention and learning deficits to emotional and behavioral issues. According to Silo Neuro Dynamic, studies have consistently shown improved cognitive functioning, reduced emotional distress, and better social interactions among patients using the device. Additionally, Myatt Att has been shown to enhance sleep quality, which is a key aspect of overall well-being for individuals with NF1.
Reducing Cognitive Deficits and Emotional Distress
One major implication of NF1 is its effect on cognitive abilities. NF1 patients often experience difficulties with focus and attention, making academic and job-related responsibilities challenging. Research has demonstrated that Myatt Att effectively reduces symptoms of cognitive deficits by targeting abnormal brain activity patterns directly. Mental health is also a particularly sensitive issue for many NF1 patients, with symptoms of anxiety, depression, and irritability common.
Explaining the improving effect of Myatt Att, Dr. Anthony Evans, another member of the research team, said, "By using the device regularly and receiving consistent therapy, we can slowly begin to change the brain's communication patterns through adjustments, breaking the patterns that are causing periods of mental distress."
The Myatt Att Treatment Plan
The treatment process with Myatt Att involves several major steps - initial assessment, personalized treatment development, and regular progress monitoring. Firstly, patients undergo thorough individualized assessments to understand their specific needs. The Myatt Att care team then creates a custom treatment plan based on this information. Each treatment lasts roughly 10 minutes and is provided via a dedicated app on the user's smartphone, making the treatment plan highly convenient. Adjustments to the plan can be made if needed and amplified by the clinician team's monitoring. This ongoing monitoring ensures that the treatment plan remains personalized and maximizes the benefits patients receive.
Security is guaranteed by following normal safety guidelines, mainly recommended for electro-stimulation therapy, such as overall good handle on the employed current and safe adaptation all across devices. Users must report immediately if discomfort or other unpleasant side effects occur.


Related Post

Unlock the Power of Efficiency: The Ultimate Guide to Admin Panel - Kampion

Unpacking the Complexity of Courtney's Character in Total Drama Island
Unraveling the Mystique: A Closer Look at Phish's Dynamic Setlists

EPSTEIN'S WHISPER: Bill Clinton's Role In The Darkest Chapters Exposed!

